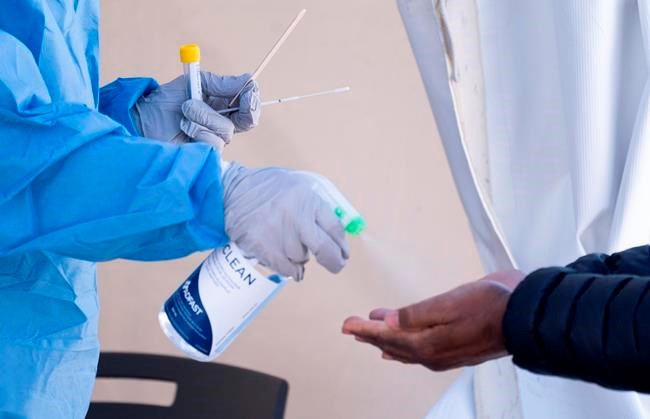
OTTAWA — Health Canada says it has authorized the first COVID-19 serological test for use in the country to detect antibodies specific to the virus.
DiaSorin, an Italian multinational biotechnology company, had developed the LIAISON test that was also recently approved by the U.S. Food and Drug Administration.
In a statement Tuesday, Health Canada says it will be used in Canadian laboratories to detect COVID-19 antibodies and help contribute to a better understanding of whether people who have been infected are immune to the virus.
Health Canada says further research will also help understand the relationship between positive antibody tests and protection against reinfection.
The Canadian agency says at least one million Canadian blood samples will be collected and tested over the next two years to track the virus in the general population and in specific groups at greater risk of having been infected, including health-care workers and seniors.
It says the testing will help inform public health decisions to protect the health of Canadians.
"Health Canada will continue to focus on the health and safety of Canadians while expediting supply of safe and effective health products related to COVID-19," the statement says.
This report by The Canadian Press was first published May 12, 2020
The Canadian Press